 English
English

Views: 0 Author: Site Editor Publish Time: 2026-03-03 Origin: Site











(VHP Generator)
I.Core Principles of VHP Sterilization
Broad-Spectrum Sterilization, Non-Toxic and Residue-Free
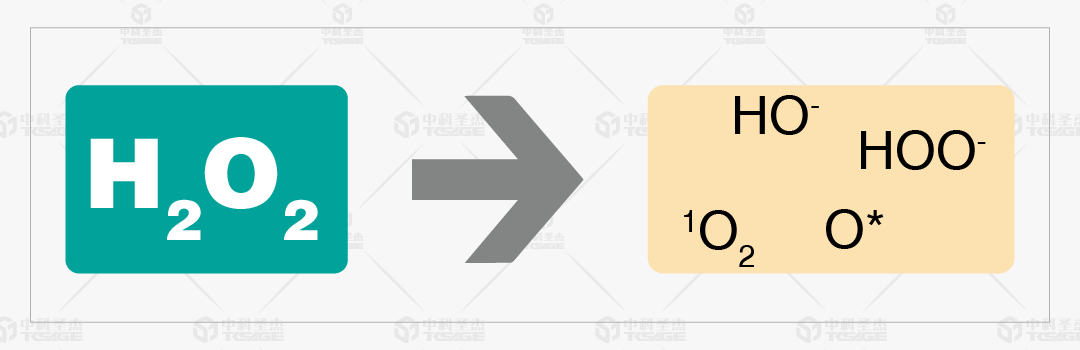
VHP sterilization works through complex chemical reactions that dissociate highly active hydroxyl and oxygen free radicals. These radicals attack cellular components, including disrupting cell membranes, lipids, and proteins, altering membrane permeability, and damaging the cytoskeleton structure. They can also enter the cell and act on DNA phosphodiester bonds, causing them to break.
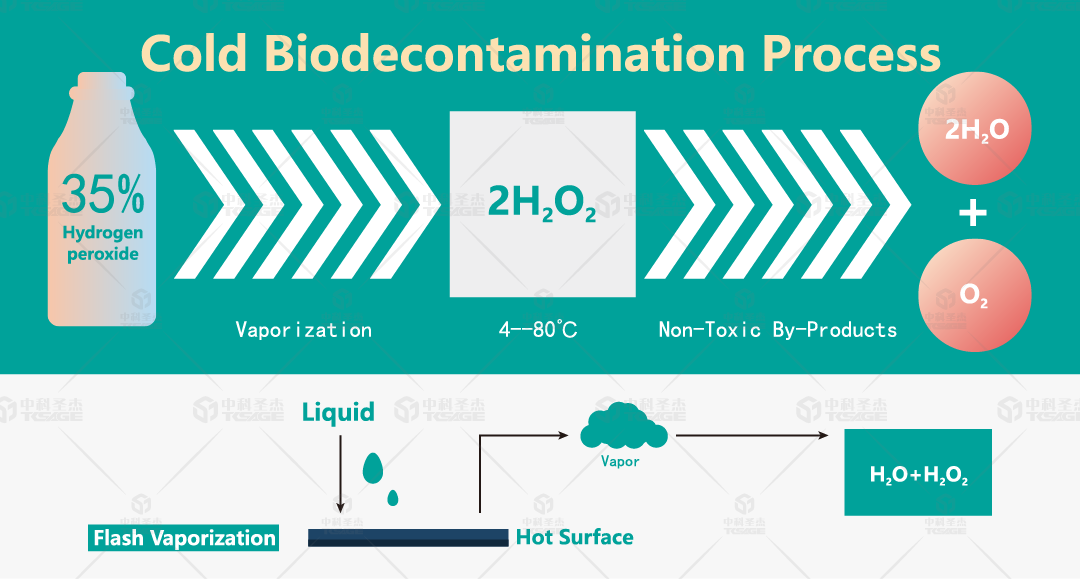
1. VHP technology involves vaporizing liquid hydrogen peroxide into a vapor state and using this vaporized hydrogen peroxide for low-temperature surface sterilization.
2. VHP has broad-spectrum microbicidal properties, effectively killing all types of microorganisms including bacteria, fungi, molds, viruses, and bacterial spores. Currently, the microorganism most resistant to VHP is Geobacillus stearothermophilus, which is therefore used as the biological indicator for VHP sterilization validation.
3. VHP sterilization is non-toxic and leaves no residue. Vaporized hydrogen peroxide rapidly kills microorganisms during the process and quickly degrades into H2O and O2 afterwards, leaving no toxic residue. The residual concentration of hydrogen peroxide is detectable.
4. The efficacy of VHP sterilization can be validated. A standard validation cycle includes parameter development, VHP distribution studies, biological challenge testing, and aeration degradation studies.
II. Comparison of Two Sterilants
Hydrogen Peroxide VS Peracetic Acid
Hydrogen peroxide (H₂O₂) and peracetic acid (CH₃COOOH) are both classified as oxidizing disinfectants. Their core bactericidal mechanism involves releasing "reactive oxygen" to disrupt critical microbial structures such as proteins and nucleic acids. However, they exhibit significant differences in chemical properties, sterilization efficacy, applicable scenarios, and safety.
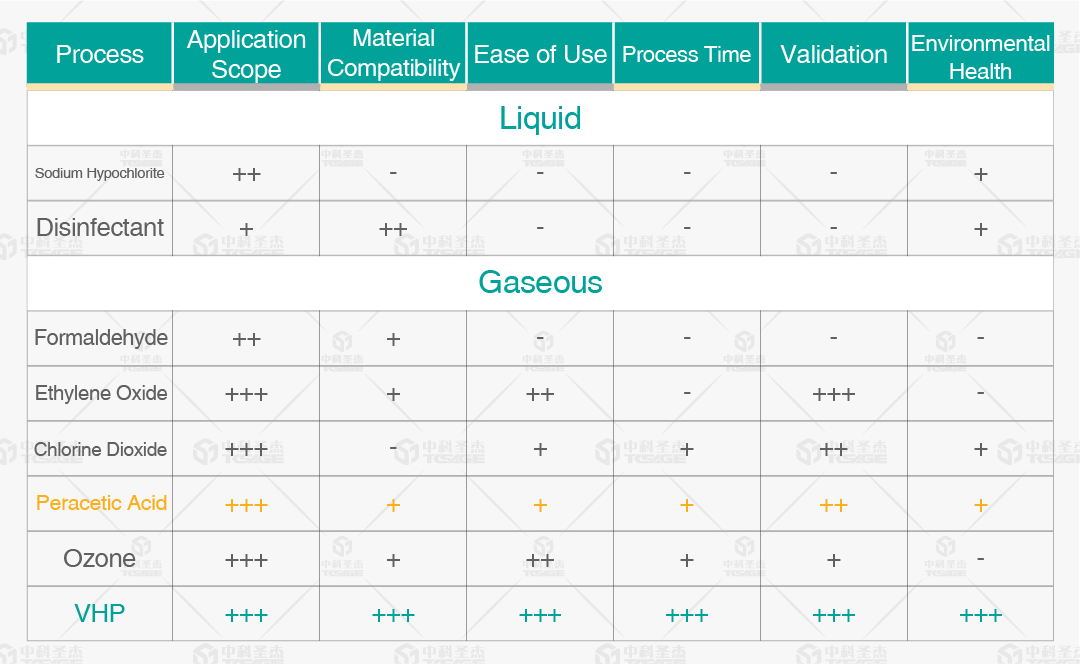
A. Bactericidal Efficacy
Bactericidal Efficacy: Peracetic acid demonstrates stronger sporicidal efficacy, rapidly inactivating spores even at low concentrations, whereas hydrogen peroxide requires higher concentrations and prolonged exposure.
B. Applicable Scenarios
Applicable Scenarios: Hydrogen peroxide is residue-free and less corrosive, making it suitable for sensitive materials (rubber, electronic components), food, and routine medical settings. Peracetic acid is suitable for terminal disinfection, cold chain/low-temperature environments, and heavily contaminated scenarios.
C. Safety
Safety: Hydrogen peroxide has low toxicity and low irritation, degrading into water and oxygen. Peracetic acid is strongly acidic, highly irritating, leaves acetic acid residue, and requires strict protective measures.
D. Stability
Stability: Both are prone to decomposition. Peracetic acid is less stable and often requires preparation immediately before use.
III. Comparison of Dry and Wet Sterilization
Low Humidity vs. High Humidity
Both dry and wet sterilization are based on the oxidative sterilization principle of vaporized hydrogen peroxide. The core difference lies in the environmental humidity requirement during sterilization, making them suitable for different scenarios and items.
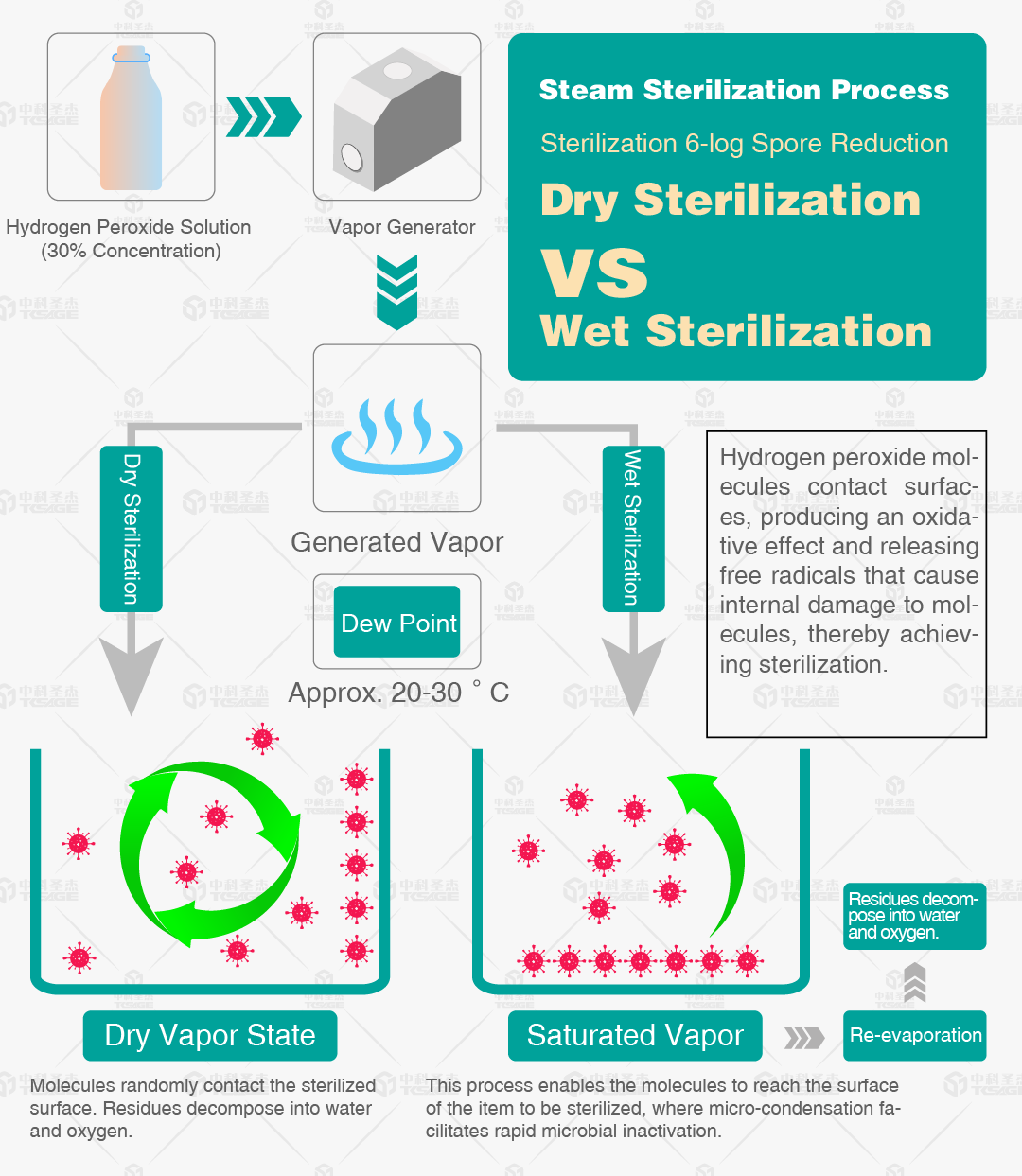
A. Dry VHP Sterilization (Low Humidity VHP)
Core Feature: During sterilization, the environmental relative humidity is controlled between 30%-50%, and hydrogen peroxide acts in a purely gaseous form.
Key Advantage: Good compatibility with moisture-sensitive items; prevents internal condensation in equipment, avoiding short circuits or component damage.
Applicable Scenarios:Sterilization of electronic equipment, semiconductor components, precision instruments, and other humidity-sensitive items.
B. Wet VHP Sterilization (High Humidity VHP)
Core Feature:Environmental relative humidity is maintained between 60%-80%, with humidity assisting the diffusion of gaseous hydrogen peroxide and microbial killing.
Key Advantage: Higher sterilization efficiency, especially against resistant microorganisms like spores, and stronger penetration, suitable for items with complex structures.
Applicable Scenarios: Medical devices, pharmaceutical cleanrooms, biological laboratories, and other scenarios where humidity is not a concern but strong sterilization is required.
C. Summary of Core Differences
Humidity Control: Dry Sterilization: 30%-50% RH; Wet Sterilization: 60%-80% RH.
Sterilization Efficiency: Wet sterilization, is slightly higher than dry sterilization, especially for highly resistant microorganisms.
Item Compatibility: Dry sterilization, is suitable for moisture-sensitive equipment; Wet sterilization is suitable for moisture-tolerant items and spaces.
Operational Requirements: The wet sterilization requires higher precision in humidity control for sealed spaces.
IV. Application Control Logic of Sterilization Technology
Temperature, Humidity, and Saturation
Humidity: Assuming an initial humidity of 40% and temperature of 25°C, the moisture content is 9.1 g/m³. With a dosing amount of 20 g/m³, post-sterilization, the water content per cubic meter becomes 9.1 + 20 = 29.1 g/m³, exceeding the saturated moisture content of 22.8 g/m³, which will inevitably cause condensation.
Saturation: In VHP sterilization technology, it refers to the saturation level of the mixed vapor of H2O and H2O2 molecules. When only water vapor is present, saturation equals relative humidity. Saturation reaching 100% will cause condensation.
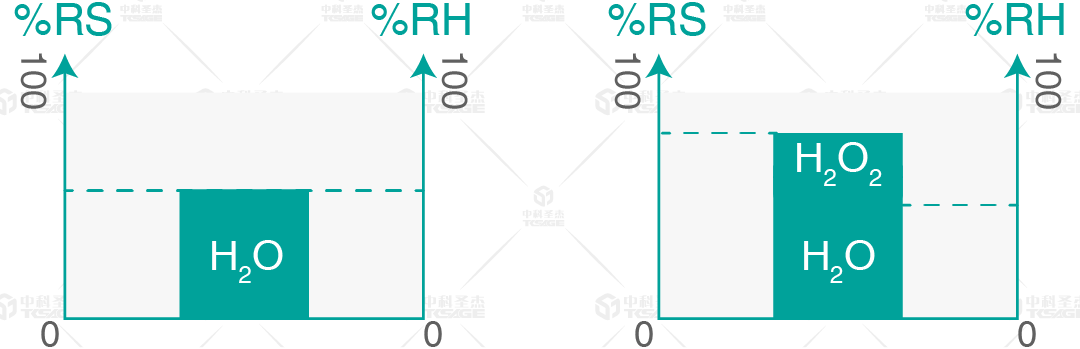
The above diagram shows two different spaces: the left one without H2O2 vapor, the right one with H2O2 vapor. When H2O2 vapor is absent, relative saturation equals relative humidity, as seen on the left. On the right, H2O2 vapor is injected into the same volume of air. Now, the relative saturation is higher than the relative humidity.
Relative Saturation (Relative Humidity) RH
The ratio of the water vapor (H₂O) partial pressure PWH₂O to the saturation vapor pressure PWS(t).
%RH=100xPWH20/PWS(t)
Relative Mixed Saturation (Relative Mixed Humidity) RS
The ratio of the mixed vapor (H₂O+H₂O₂) partial pressure PWH₂O+H₂O₂ to the saturation vapor pressure PWS(t).
%RS=100xPWH20+H2O2/PWS(t)
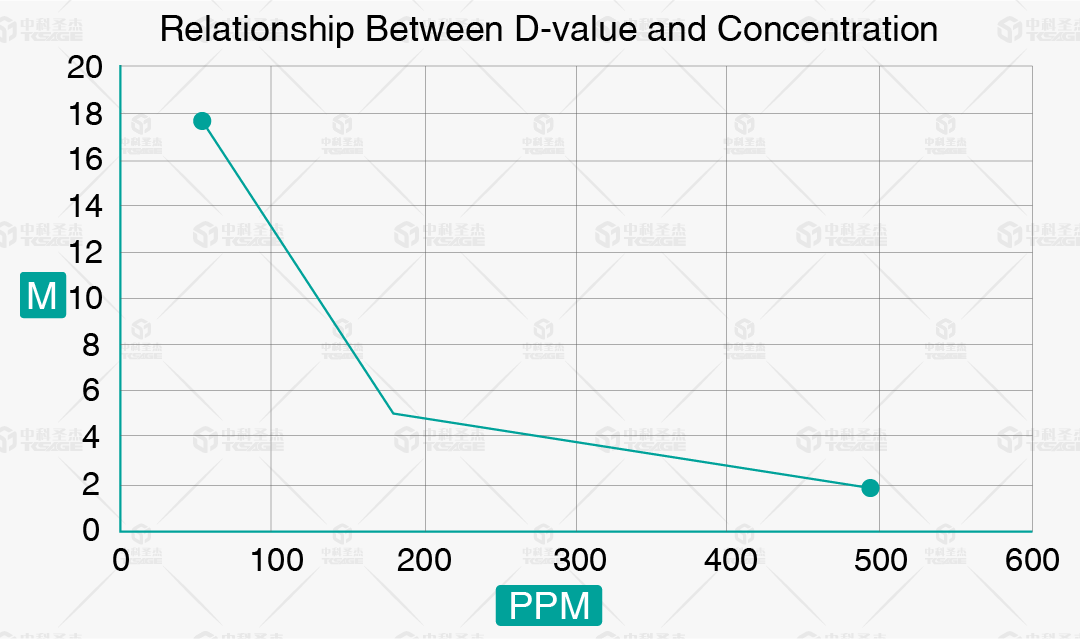
(VHP Sterilization D-value Curve)
S-value: Under standard atmospheric pressure, the vapor relative saturation during the sterilization process calculated based on VHP concentration, relative humidity, and temperature.
D-value: The time required to reduce the microbial population by 1 log (90%).
F(D)-value: The integral of the D-value over time during the sterilization process. A target LOG reduction value can be set, and the corresponding sterilization effect is achieved by integrating the F(D) value over the process.
The calculated F(D) value is compared with the set F(D) value to determine whether adjustments to the chamber environment are needed (e.g., continue injecting hydrogen peroxide, end sterilization, etc.). Sterilization ends when the calculated F(D) value is greater than or equal to the set F(D) value.
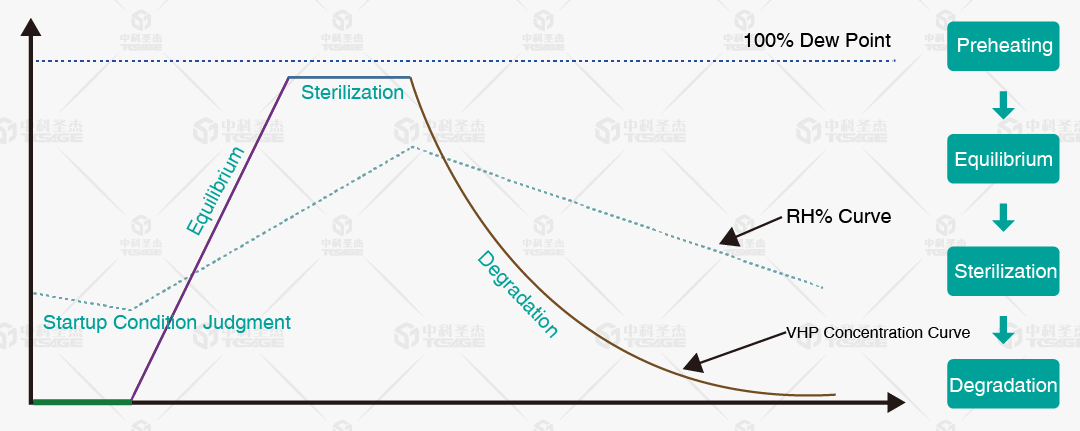
(Sterilization Process Schematic)
Saturation Control: During sterilization, the medium is a mixed vapor of H2O and H2O2. Ensure the mixed vapor saturation remains below the dew point.
V. VHP Sterilization Applications
Biopharmaceuticals, Biosafety Laboratories, Healthcare

(Application of VHP Sterilization Technology on Products Schematic)
Application in the Biopharmaceutical Industry: The biopharmaceutical industry requires a "sterile environment/materials" throughout the entire production process (from cell culture to finished product filling). Any microbial contamination may lead to batch failure or product safety risks. VHP technology has become a critical sterilization method as it meets the requirements of being "highly effective, residue-free, and non-damaging to materials."
Application in Biosafety Laboratories (BSL): Biosafety laboratories (especially BSL-3/BSL-4 levels) primarily handle highly pathogenic microorganisms (such as SARS-CoV-2, Ebola virus, Mycobacterium tuberculosis, etc.). Strict sterilization is required to prevent "microbial leakage" (protecting personnel and the external environment) and "cross-contamination" (protecting experimental samples). VHP technology is a core method for laboratory "terminal sterilization" and "localized sterilization."
Application in the Healthcare Field: Sterilization needs in healthcare focus on "reuse of medical devices" and "disinfection of clinical environments," particularly addressing pain points such as "thermosensitive medical devices that cannot withstand high-temperature sterilization" and "incomplete terminal disinfection of infectious disease wards." VHP technology demonstrates unique advantages in these scenarios.