 English
English

Views: 0 Author: Site Editor Publish Time: 2026-04-07 Origin: Site









As a core protective device in biological laboratories, biological safety cabinets play a critical role in tasks such as pathogenic microorganism detection and biological sample processing. They protect laboratory personnel, experimental samples, and the surrounding environment from contamination, serving as an essential barrier within the laboratory biosafety system.

During practical operation and daily operation, influenced by factors such as operating procedures, equipment maintenance, and environmental conditions, biological safety cabinets often experience issues like airflow anomalies, inadequate disinfection, operational errors, and equipment malfunctions. These problems can not only reduce protective effectiveness but also pose biosafety risks. To standardize the usage procedures of biological safety cabinets, promptly identify and resolve common issues, and ensure the safe and orderly conduct of experimental activities, this article, based on practical application scenarios, outlines typical problems encountered during use and proposes corresponding solutions. It serves as a reference for the safety management and standardized operation of biological safety cabinets.
Protecting Operators and Experimental Materials

The safety cabinet features a front access opening through which operators can perform work inside the cabinet. Inward negative pressure airflow at the front access opening protects the operator's safety. Downward airflow, filtered through a ULPA filter, protects experimental materials inside the cabinet. Air is exhausted from the cabinet after passing through a ULPA filter to protect the environment. The airflow pattern is free of dead angles, vortices, or outward escaping airflow.
The safety cabinet provides an optimal clean and physical containment barrier for airborne biological or particulate chemical agents, reducing risks (including contamination risks to personnel, materials, and the environment) during operations conducted within it. However, to ensure the safety of personnel, products, and the environment, safety cabinet operators must undergo professional training. Only after a thorough understanding of safe operating procedures can relevant experimental operations be conducted inside. Improper use of a safety cabinet can directly harm products, operators, and the environment due to biological contamination.
Standard Operating Methods and Precautions
During operation, maintain the front window opening height at 200 mm (ensuring the inflow velocity at the access opening). This airflow barrier is critical for containing hazardous materials within the cabinet. Do not block the front intake grille with your arms, paper, film, or laboratory equipment.
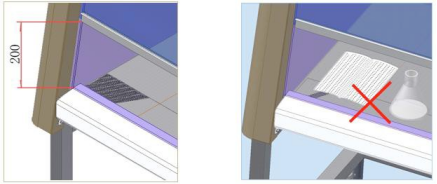
Warning: Ensure the front window opening height is correct during operation.
Warning: Prevent obstruction of the front intake grille.
Electrical Safety Precautions for Safety Cabinet Use: Due to the specific biosafety nature of the cabinet, if internal electrical circuit maintenance (such as fan or sensor line replacement) inside the cabinet body (excluding the electrical panel) is required, a biological hazard assessment must be conducted first, and the work should then be handled by qualified professionals.

If the safety cabinet needs to be installed or used in conjunction with other medical devices, please contact a biosafety professional or the manufacturer for assessment first to confirm that the other medical devices will not affect the protective airflow of the safety cabinet.
Safety cabinet operators must receive training on safe operation, biological hazards, electrical safety, and fire safety. Only after fully understanding the potential hazards can they operate the equipment.
Cause Analysis and Control Methods
Product Contamination |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
Hazards and Countermeasures
| |
| |
| |
| |
| |
| |
| |
| |
Countermeasures: |
| |
| |
| |
| |
| |
| |
Product Strength / After-Sales Service / Customization Support

What are the practical advantages of the structure and materials?
The external casing is made of high-quality cold-rolled steel with electrostatic spray coating. The three-sided work area is constructed from integrally formed SUS304 stainless steel with large-radius rounded corners, eliminating cleaning dead spots and resisting dirt accumulation and corrosion, facilitating easy disinfection and wiping.
What are the features of the airflow system and safety protection?
Cigeair offers Class II, Type A2 (70% recirculation / 30% exhaust) and Type B2 (100% full exhaust) models. They utilize a vertical laminar flow negative pressure design, ensuring triple protection for personnel, samples, and the environment. The front window air intake is designed to prevent reverse flow, enhancing operational safety.
What intelligent control and alarm functions are available?
Equipped with an LCD color touchscreen interface, displaying real-time airflow velocity, filter status, and operating duration. Features audible and visual alarms for filter clogging/failure, fan overload, and abnormal window sash conditions. Supports parameter memory and power failure recovery.
What are the company's manufacturing and service advantages?
The company boasts a 35,000 m² production base and a 2,000 m² R&D center, integrating R&D, production, installation, and after-sales service. A professional technical team provides full-process services. The equipment is suitable for applications in biopharmaceuticals, clinical laboratories, and scientific research facilities.
What is the after-sales service system for Cigeair Biological Safety Cabinets?
Cigeair has a 24-hour national after-sales service hotline, providing round-the-clock fault response. It offers a complete closed-loop service covering installation, commissioning, training, and maintenance. Supports on-site repair, filter replacement, performance calibration, and other after-sales activities to ensure long-term stable equipment operation.
Is customization support available?
Yes, customization is available on demand. Cabinet dimensions, access opening height, and internal layout can be adjusted based on laboratory space and usage scenarios. Custom interfaces, power configurations, and exhaust connection solutions can be provided. Non-standard structural optimization can be performed in accordance with GMP/laboratory specifications.
What guarantees are provided for equipment installation and operation training?
Cigeair provides free on-site installation and commissioning services, including on-site performance verification and compliance acceptance. Technical personnel are arranged to provide specialized training on operating procedures, routine maintenance, and alarm handling. A full set of operation manuals and maintenance documentation is delivered simultaneously.
Can a complete laboratory integration solution be provided?
Cigeair can provide an integrated solution combining biological safety cabinets with a clean environment, connecting pass-through chambers, laminar flow hoods, negative pressure weighing stations, VHP sterilization equipment, etc., to create a compliant clean laboratory. This solution is adaptable to various scenarios, including hospitals, disease control centers, pharmaceutical companies, and research institutes.